Public Health Advocacy
Yesterday, the Georgia House approved HB 80, the amended FY21 budget that runs through June 30, by a vote of 149 - 20. This bill included >$33M in new state spending for public health. Those funds will cover the addition of three new senior level positions, $18M to replace the GRITS immunization registry (dates back to 1996) and $15M for the ADAP program that provides critical HIV medications to those who cannot afford them. The bill moved to the Senate Appropriations Committees today for their consideration. I’m hopeful that the Senate subcommittee that oversees the Department of Public Health will add more funds. Please consider contacting the members of that committee as well as your state senators. You can look up your state senator here and the senate appropriations subcommittee (with contact info) here.
I don’t mean to seem ungrateful, $33M is a lot of money. And these are critical programs that needed funding. But these are band-aids to plug just two holes that DPH faces. They are also not the kind of investment that will help the rank and file members of the Department of Public Health with their staffing shortages or lack of resources. It is not enough to say we’ll address that in the FY22 budget (that goes into effect on 01Jul) because the DPH staff are overwhelmed now. Earlier this week, an article in the Atlanta Magazine identified that DPH staff were under a 90 day moratorium for taking annual leave through April 16th. I was able to confirm this with a DPH employee too. And I’m sure similar things are happening at public health districts and county health departments as well.
This seems to run contrary to the Commissioner’s comments before the House Appropriations Subcommittee when she indicated that to improve morale at DPH, the employees would need a simple “thank you” rather than money. To be fair, Dr. Toomey is a political appointee and public health is inherently political. It’s quite possible that she wants to advocate for her people but can’t due to the political landscape. But these public health professionals, who have been working nonstop for almost a year now and face significant burnout, are not allowed to take leave that they’ve earned. In public health, people are one of the most valuable resources we have. And they are burned out. These people have children and families and friends who need them too. They have medical issues of their own. They have their own mental health concerns after working nonstop for a year with an ever growing workload. More than anything though, it reinforces the idea that the DPH staff are understaffed and under-resourced. So it really is important that we contact and implore the senate subcommittee to add additional DPH funds to the amended FY21 budget and continue that commitment into the FY22 budget too.
So yes, we have a small victory to celebrate here. But the work is not done to ensure that the DPH, public health districts and county health departments have the resources they need to cope not only with this pandemic but the other pressing issues of public health concern that might be neglected right now.
Vaccine Update
Today’s newsletter is going to focus a lot on various pieces of vaccine news since Georgia’s COVID-19 numbers are sort of more of the same right now.
New Vaccines
There are two vaccines that are getting closer to emergency use authorization in the US from Oxford-AstraZeneca and Johnson and Johnson (Janssen). The New York Times has some excellent tutorials with diagrams on how the AstraZeneca and Johnson and Johnson vaccines work and I will refer you to those.
These are different from the Pfizer and Moderna vaccines that are sort of the “minimalist” versions of a vaccine - they are made of short-lived RNA with very few moving parts. I think we’ve all seen examples that simpler machines, systems, etc, have fewer things that can go wrong. So it might not be surprising that the RNA vaccines have demonstrated really good safety data both in clinical trials and since their use for the public (more on that in a minute). The AztraZeneca and Johnson and Johnson vaccines are DNA vaccines where the SARS-CoV-2 (remember, this is the virus that causes COVID-19 disease) gene for the spike protein is inserted into an adenovirus genome (also made of DNA). This is a different family of viruses than coronaviruses, many of which can cause the common cold. The adenovirus is a “vector,” a relatively inert delivery vehicle for the important SARS-CoV-2 viral gene that codes for the spike protein. It has been engineered so that it can enter your cells like a regular adenovirus, but cannot replicate itself once its inside. That means these are dead end viruses. For that reason, they cannot give you an infection. But the DNA inside them can be transcribed (making RNA from the DNA) and translated (making protein from the RNA). One difference here with the RNA vaccines is that the enzymes in your cell that do the DNA -> RNA conversion are in the nucleus of the cell (where your DNA is held). So whereas the Pfizer and Moderna vaccines bypass the nucleus altogether, the AstraZeneca and Johnson and Johnson ones *do* need to enter the nucleus to kick off protein production. From there, the spike protein-encoding DNA is transcribed into mRNA (the same kind of molecule that’s in the Pfizer and Moderna vaccines) and then the mRNA leaves the nucleus to meet up with a ribosome (protein builder) that will read the instructions in the mRNA to make the spike protein. Those proteins are expressed on the surface of your cells for your immune system to see and react. From thereon, the immune system responds the same way it would to the Pfizer or Moderna vaccines.
Because these new vaccines are made of DNA, they’re more stable than the RNA vaccines. They don’t require freezer or ultracold storage. Instead, they can be held in a standard refrigerator for months. So these will be especially helpful for reaching rural and medically underserved populations. Whereas the mRNA vaccines are brand new platforms for vaccines, the adenovirus/DNA vaccines have been studied for decades in developing vaccines for HIV, Zika, and Ebola, especially by Johnson and Johnson.
Today we got an update regarding efficacy and safety for the Johnson and Johnson vaccine. In the US, the vaccine had a 72% efficacy (66% globally) in preventing moderate and severe COVID-19 disease 28 days after the dose. After 28 days it displayed 85% efficacy in preventing severe cases and 100% efficacy in preventing death. After 49 days it protected against severe disease at an efficacy of 100%. This includes protection against the South African variant of SARS-CoV-2.
On it’s face, it might seem like a disappointment to only see 72% efficacy in the US 28 days after the vaccination, especially when we’re seeing 95% from the RNA vaccines. Let’s keep something in mind though. The RNA vaccines only achieve 95% efficacy after a second dose whereas the Johnson and Johnson vaccine is a single dose regimen. That means you don’t have to schedule a follow up dose. And, the results after 28 and 49 days are pretty fantastic, maybe even comparable to what we see from the RNA vaccines.
Moving on to the AztraZeneca vaccine. The European Union authorized this vaccine for use in all adults today. It is a two-dose series spaced 4-12 weeks apart. That may give more flexibility in scheduling the second dose if resources are scarce. However, countries like Germany are concerned that there might not be enough data to indicate that it works well in adults >65 years of age who have waning immune systems as a function of normal human aging. Based on data presented by the Advisory Committee on Immunization Practices (ACIP) yesterday for the US Phase III clinical trial, just under 24% of trial participants in the US are over age 65. So there are some data, but maybe not enough yet for the Germans. In any event, it will help to ease the supply problem, even if countries like Germany do prioritize these vaccines for younger adult populations. What I really like about the study plan for this vaccine is how diverse their study population is, including several different countries, including all three countries where variants have emerged - the UK, Brazil and South Africa. Like the Johnson and Johnson vaccine, this vaccine demonstrates reduced efficacy in the short term compared to the RNA vaccines - 70.4% efficacy in preventing symptomatic COVID-19 infection. But there were no hospitalizations or deaths after 21 days post-vaccination. So that’s similar to the Johnson and Johnson vaccine and, really, pretty great. The vaccine also shows remarkably good safety data so far, with only 4 severe adverse reactions that were potentially attributable to the injection - 2 in the vaccine group and 2 in the placebo - out of tens of thousands of trial participants. Like the Johnson and Johnson vaccine, the AstraZeneca vaccine can be held in standard refrigerator conditions for up to 6 months.
SARS-CoV-2 Variants and Vaccines
An issue of considerable concern is how well our existing vaccines will do at protecting against the emerging variants from the UK, Brazil and South Africa. The South African variant is thought to have the biggest potential impact on the efficacy of monoclonal antibody therapy (the kind of pre-made antibodies that President Trump received during his infection). Unfortunately, the South African variant was reportedly detected yesterday in South Carolina. However, the US has lagged behind other nations when it comes to conducting genomic surveillance for these variants. Yesterday, the ACIP gave an update on how the US is leveraging available resources to scale up genomic surveillance (see below).
So right now there is capacity to sequence and analyze at least 2500 viral specimens per week. There’s possibly more being done at universities, but they don’t give an estimate. For reference, the US 7-day average for new cases is >159,000. So we are testing an extremely small sample size. Without a robust surveillance system, it’s likely that the variant has been here for some time. The more this virus spreads, the more opportunity it has to mutate. We are relatively lucky for now that the variants are not more lethal than the wild type virus. But we still need to contend with the reality that the variants are more transmissible and/or not as well-controlled using monoclonal antibody therapy. If we want to prevent further mutations from happening, we need to do more to limit transmission. In the meantime, Moderna announced they are producing a “booster” version of their vaccine that would cover the South African variant, just in case it is needed. The cool thing about the RNA vaccines is that they are easier to update quickly, swapping out one piece of RNA for another.
Do the vaccines limit transmission?
The primary goal of the vaccine clinical trials has been to assess whether they prevent severe COVID-19 disease. And so far they are really good at that. But determining whether the vaccine also prevented asymptomatic infection or transmission to others was not a focus of the trials. This is part of the basis for why we’re telling people they need to continue to wear masks and socially distance themselves, even after vaccination. There’s a good chance that it does interrupt transmission, but we need to know for sure, especially when disease prevalence is so high. The ACIP meeting yesterday also provided some update in this area. As you can see, it really was an important meeting with a lot of data.
They are studying a cohort of >5000 healthcare workers who were among the first to be vaccinated. These individuals are tested weekly for the presence of SARS-CoV-2 and they are further studying any secondary transmissions to household contacts of these healthcare workers if and when they are sick or exposed to COVID-19 following vaccination. This study is running through March 2022. Yes, more than a year from now. We will likely have some interim results before then, however.
They are also expanding to study members of the general public now that eligibility for vaccination has expanded.
Vaccines being tested for Children
I don’t know about you. But for all this talk of “when will we be back to normal,” as a parent I don’t think I will behave like normal until my children are also vaccinated for COVID-19. So it was neat to get an update from ACIP yesterday on the studies that are ongoing to assess safety and efficacy of the vaccines for children.
Teenagers are enrolled and it appears that studies are ongoing. For younger children, it seems that we are still in the very earliest stages. However, there is more promise for the Janssen (this is part of Johnson and Johnson) vaccine since this type of vaccine has previously been studied and used in teens, infants and children. Hopefully we’ll learn more in coming weeks.
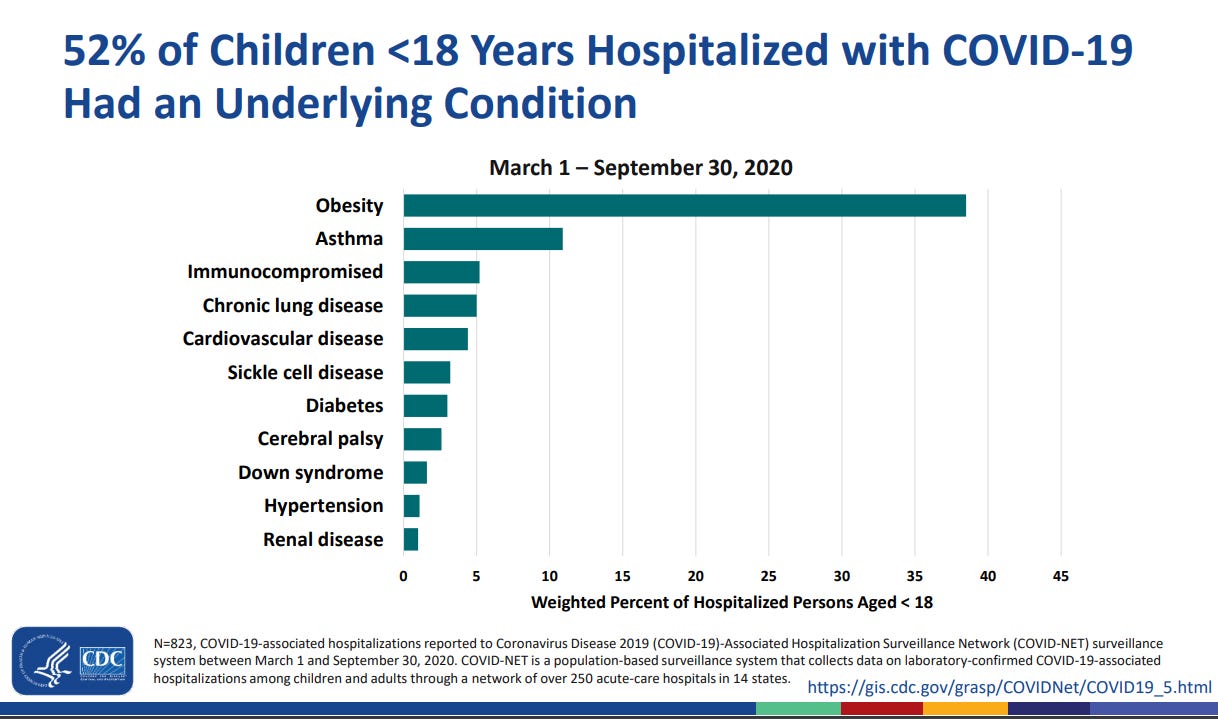
This next presentation from ACIP wasn’t really vaccine focused but talked about severity of illness among children infected with COVID-19. I thought I would bring this graphic to the newsletter because we haven’t talked about these data in a while. For children who have been hospitalized with COVID-19, only 52% of them had an underlying condition. That means that 48% were healthy kids prior to COVID-19. The number one underlying condition associated with hospitalization (and by a large margin) was obesity. It’s important to remember that a child can have more than one of these conditions at once. In addition, these are just the children admitted in states and hospitals that are part of COVID-NET, a surveillance network consisting of 14 states. So it is a good estimate, but may not be a perfect representation of the national situation.
The other slide that I thought was really interesting was this next one, where they estimate the infection rates per 100,000 for each age group, adjusting for under detection (i.e. due to poor testing or the number of asymptomatic infections). These estimates suggest that K-12 aged children have an infection rate that is higher than 50-64 year olds and not that far off from the 18-49 year old rate. Infection rates among 0-4 year olds are estimated to be comparable to those 65+. Other data in the presentation go on to demonstrate that children are less likely to require hospitalization or to experience death, which is not new information but important to remember. But I think this graph from CDC shows that it is a myth to purport that children somehow don’t catch COVID-19 as easily as adults. It’s all the more reason for them to take the same precautions we are recommending for adults including wearing masks whenever indoors with non-household contacts and remaining socially distant.
Georgia Numbers
Testing
It was a robust day for testing with 40,314 PCR tests reported through the Electronic Laboratory Reporting system. But due to some lower output earlier in the week, I think it’s likely we are going to see a decrease in weekly output when I do that analysis on Sunday. In the meantime, 13% of today’s PCR tests were positive. That’s the lowest number we’ve seen in a while. Antigen testing identified 27% of today’s newly reported cases, but we don’t get data on total antigen tests performed nor how many were positive from the state.
Cases
Today there was a net increase of 6558 newly reported cases (4786 by PCR, 1772 by antigen test). The 7-day case rate per 100,000 continues to fall. As of today, the state case rate is 64% above the summer surge peak but 39% less than the winter peak. So we still have a very high rate of disease, even as things are decreasing.
The School Aged COVID-19 Surveillance Data report for this week is due today but hasn’t yet been posted.
Hospitalizations
There were 261 new COVID-19 admissions and 29 new admissions to the ICU. There are currently 4455 patients hospitalized for COVID-19. The last time we saw a number like this was 29Dec, so about a month ago. This peak appears to have been more intense but shorter in duration than the summer one.
The concern I expressed for Hospital Region J on Wednesday appears to no longer be an issue, the patient census there is decreasing again. But something weird started happening with Hospital Region G yesterday and continues into today. As you may recall, they’ve been contending with an overage of ICU bed demand for three weeks. Then yesterday they dropped from 114% bed usage to 73%. This is mostly explained by a huge jump in the number of available beds locally - it jumped from 184 on Wednesday to 259 on Thursday. That seems….unlikely to be real. I’ve inquired with colleagues from the state’s COVID-19 data task force and they too think there’s a problem with the capacity numbers (it’s also affecting the overall state total). But the person who usually manages and checks these data is not at work and not backfilled. So I guess we’re going to have questionable data for a bit.
Assuming the region G data are correct, there are no hospital regions using >100% of their ICU beds today. Further, there is only one region (M) where >40% of admitted patients are COVID-19 patients.
Deaths
There was a net increase of 130 newly reported deaths today. Thirty three percent each came from nonrural and rural counties, respectively, outside of the Atlanta metro. The death rate remains very high for rural counties compared to all other county types.
References
https://www.atlantamagazine.com/news-culture-articles/how-chronic-underfunding-fueled-georgias-covid-19-vaccine-woes/
https://amberschmidtkephd.substack.com/p/the-daily-digest-25jan2021
https://www.nytimes.com/interactive/2020/health/johnson-johnson-covid-19-vaccine.html
https://www.nytimes.com/2021/01/28/us/covid-south-africa-variant-south-carolina.html
https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-01/02-COVID-Villafana.pdf
https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-01/10-COVID-Oliver.pdf
https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-01/09-COVID-Fleming-Dutra.pdf
https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-01/04-COVID-Erbelding.pdf
https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-01/03-COVID-Campbell.pdf
Georgia COVID-19 Updates is a free newsletter that depends on reader support. If you wish to subscribe please click the link below. There are free and paid options available.
My Ph.D. is in Medical Microbiology and Immunology. I've worked at places like Creighton University, the Centers for Disease Control & Prevention and Mercer University School of Medicine. All thoughts are my professional opinion and should not be considered medical advice.











I wonder if the ATL suburb cases are so high due to the UK variant. Cobb county has confirmed that the variant is in our county. I'm unsure about the other suburbs.